Parasitic Nematodes in Corn
June 19, 2024
Summary
- Nematodes are small, thread-like organisms that feed on corn roots.
- Nematode feeding reduces plant vigor and creates openings for pathogens.
- The presence of nematodes can limit yield potential, but nematodes can remain undetected until a field is sampled as nematode symptoms can mimic others, such as drought and nutrient deficiency.
- Management of nematodes requires an integrated approach, and the correct identification of the species is critical.
Introduction
Nematodes are microscopic, thread-like organisms which can be found in virtually any field where corn is grown. While some species of nematodes can be beneficial, other nematodes parasitize plants and may reduce yield potential by limiting a plant’s ability to take in water and nutrients.1 Nematodes can also create openings for other pathogens to enter the plant and cause disease. The effects of nematodes in a corn field can vary from causing no obvious symptoms on corn plants to causing severe injury and tremendous yield loss.2
Life Cycle and Environment
Corn nematodes spend their entire life in the soil. Nematode populations in a field may vary based on soil type, environmental conditions, and whether growing plants are present or not. Corn nematodes may flare up in continuous corn cropping systems, but populations of some nematode species can still be maintained when a field is planted to a non-host crop such as soybean or alfalfa. Many nematode species thrive in sandy soils, though corn nematodes can be found almost anywhere, including heavier soils. Under favorable conditions, most parasitic nematodes of corn can complete their lifecycle (egg, four juvenile stages, egg-producing adult) in about one month, and a population may complete four or more lifecycles in one growing season.1 Mature females of some species—such as the root-knot nematode—can produce hundreds of eggs during a lifecycle, leading to rapid population growth.
Parasitic nematodes are classified by where they live while feeding on the plant, which is important information to have when sampling for nematodes. Endoparasitic nematodes live within the root structure of the plants they parasitize, while ectoparasitic nematodes live in the soil outside of the roots.

Symptoms
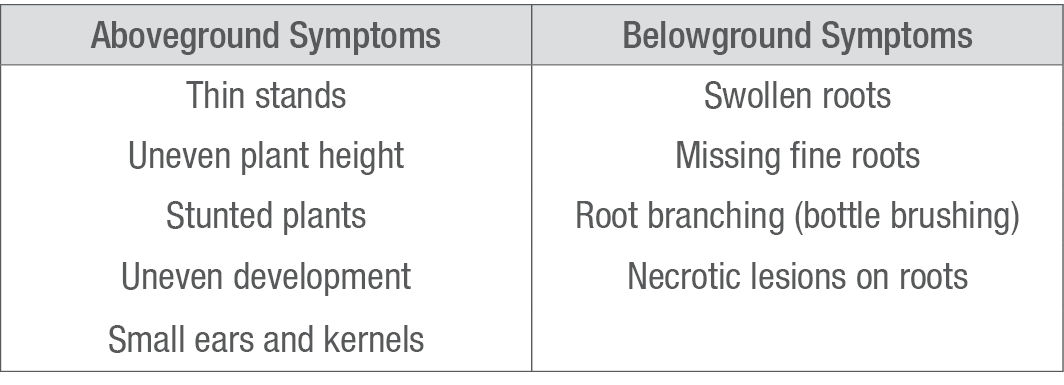
Symptoms of corn nematode feeding are typically found in irregular patches, not across a whole field (Figure 1). There are often no aboveground symptoms of nematode feeding unless population levels are extremely high, though symptoms may also intensify under stressful growing conditions. Belowground symptoms vary by nematode species (Table 1). For example, root-knot nematode infection typically causes galls (irregular swelling of the roots), while stubby-root nematodes cause stunted, blunt, and swollen roots. Foliar symptoms may be hard to identify, as they are not unique to nematode feeding and can be easily confused with nutrient deficiencies, herbicide damage, or insect damage.1 The only way to accurately diagnose the presence of parasitic nematodes in corn is through soil and root sampling.
Table 1. Common symptoms of corn nematode injury.

Nematode Species
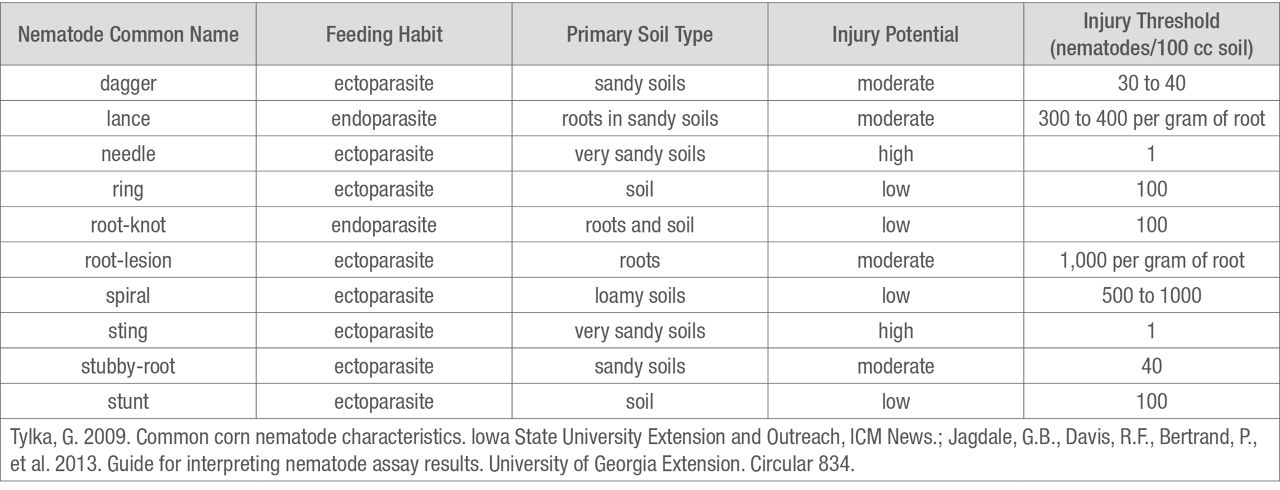
There are many different nematode species that interact with corn. They vary in size, feeding habit, damage potential, and preferred soil type (Table 2). Some nematodes can only be found in very sandy soils, while others can be found in a wide range of soil types. Listed below are nematodes that are economically important in corn production.
Lesion (Pratylenchus spp.)
Lesion nematodes are small, migratory endoparasites, and are probably the most important corn nematode in the Midwest. They can be found across a wide range of soil types. Damage from lesion nematodes can range from small water-soaked areas to severe necrosis of the roots.4 The damage threshold for lesion nematodes is 1,000 nematodes per gram of root, and populations can reach numbers of 10,000 to 84,000 nematodes. Threshold numbers may vary based on environmental conditions.5
Needle (Longidorus spp.)
Needle nematodes are among the most devastating type of nematodes in corn. They are ectoparasitic, and their presence is highly restricted to sandy soils. These nematodes damage corn roots by feeding on root tips, stunting the lateral roots, and essentially destroying the fibrous root system. Damage thresholds for needle nematodes are as low as one nematode per 100 cubic centimeters of soil; 25 nematodes per 100 cubic centimeters of soil can cause severe damage.4
Lance (Hoplolaimus spp.)
Lance nematodes are endoparasites mostly found in sandy soils, but they can be found in many soil types. They are extremely common and have a wide range of hosts; therefore, crop rotation is ineffective in controlling lance nematodes.4
Dagger (Xiphinema spp.)
Dagger nematodes are ectoparasitic and can cause stunting and chlorosis of corn plants. They are found in sandy and silty loam soils and are known for their long lifecycle. They reproduce once per year and can live up to 5 years in favorable conditions. Tillage may be effective in controlling dagger nematodes.4
Stubby-root (Paratrichodorus sp.)
This ectoparasitic nematode is a serious pathogen in southern states and is found in sandy soils.5 Stubby-root nematodes can be very damaging. Symptoms include stunted, blunt, and swollen roots due to nematode feeding on the terminal ends of young roots.
Root-knot (Meloidogyne spp.)
Root-knot nematodes are endoparasitic, remain sedentary inside the root, and cause small galls on corn roots. Soybean plants are also a host of root-knot nematode, so rotation to soybean is not an effective control method.

Sampling for Nematodes
To confirm the presence of nematodes, soil and root samples must be collected and sent to a nematode testing laboratory for analysis. It is important to collect both soil and root samples to determine the presence of endoparasitic and ectoparasitic nematodes. Sampling should occur during the middle of the growing season, when nematode numbers are greatest and damage symptoms are apparent.1,2,3 Soil samples should be taken around the edge of symptomatic areas, at about 12 inches deep, within a few inches of the damaged plants (or intersecting the root zone). The soil should not be overly wet or dry when sampling, and at least 20 soil cores should be collected per sampling area (10 acres or less).3 Roots from two to three corn plants should also be collected. Root and soil samples should be refrigerated until the time of shipping.
A general field survey may also be conducted to determine the presence and potential risk of nematodes in a field. This is commonly done by taking soil samples post-harvest in fields where no damage symptoms have been observed. Additional information and instruction on nematode sampling can be found from your local Extension service or nematode testing laboratory.
Management
Once corn nematodes are present in a field, management practices should be used to help minimize crop damage and keep population densities low. Because there are many nematode species, identification is essential for determining the appropriate control option. For certain nematode populations, the best management practice is crop rotation. Corn management practices that reduce crop stress may also help the crop overcome nematode attacks. Additionally, the following agronomic practices may help farmers manage potential nematode infestation:
- Fertilize – Plants suffering from nutrient deficiency are more susceptible to injury.
- Weed Control – Weeds are hosts for many nematodes; managing weeds can help keep nematode populations low.
- Crop Rotation – For certain nematode species, rotating to a non-host crop can keep population densities low.
- Chemical Control – Nematicides and seed treatments may be an effective control measure against corn nematodes.
Nematode management requires an integrated approach and is an ongoing process. Sampling and identification of the nematodes present is crucial for creating an effective management strategy against corn nematodes.
Table 2. Common corn nematode characteristics and injury thresholds6,7
Sources
1Grabau, Z.J. and Vann, C. 2021. Management of plant-parasitic nematodes in Florida field corn production. University of Florida. ENY-001. https://edis.ifas.ufl.edu/publication/NG014#TOP
2Jackson, T. and Timmerman, A. 2011. Sampling for nematodes in corn. University of Nebraska–Lincoln, CropWatch. https://cropwatch.unl.edu/sampling-nematodes-corn
3Tylka, G. 2007. Nematodes in corn production: A growing problem? Iowa State University Extension, ICM News Archive. IC-498. https://crops.extension.iastate.edu/encyclopedia/nematodes-corn-production-growing-problem
4Harbach, C. and Kleczewski, N. 2022. Nematodes. In, Illinois agronomy handbook. University of Illinois Extension. https://extension.illinois.edu/sites/default/files/nematodes_2022web.pdf
5Norton, D.C. and Nyvall, R.F. 2011. Nematodes that attack corn in Iowa. Iowa State University Extension and Outreach. PM1027. https://store.extension.iastate.edu/product/4357
6Tylka, G. 2009. Common corn nematode characteristics. Iowa State University Extension and Outreach, ICM News. https://crops.extension.iastate.edu/cropnews/2009/08/common-corn-nematode-characteristics
7Jagdale, G.B., Davis, R.F., Bertrand, P., et al. 2013. Guide for interpreting nematode assay results. University of Georgia Extension. Circular 834. https://extension.uga.edu/publications/detail.html?number=C834&title=guide-for-interpreting-nematode-assay-results
1211_119748